|
Two recent studies showed increased RBD erectability and enhanced receptor binding by the Alpha and Beta spikes 24, 25. This enabled us to directly compare the pre-fusion spikes of the new variants with those of the original strain (first identified in Wuhan) 5, 8 and the D614G-only variant 12 we described in previous studies, findings that agree with reports that used non-stabilised spikes 6, 7, 13, 23. 
We used pre-fusion-stabilised spikes 22 which have been established as a tool to study the receptor-binding properties in the absence of the conformational change associated with fusion. Here we have examined the structures and receptor-binding properties of spikes of the B.1.1.7 variant first described in Kent, United Kingdom (now termed Alpha) 10, 16, 17, 18, 19 and the B.1.351 variant first described in South Africa (now termed Beta) 20, 21. Recently emerging variants of SARS-CoV-2 have acquired other substitutions in the spike including a number located at the monomer-monomer interfaces, at the receptor-binding site, and near the furin-cleavage site (Supplementary Fig. The D614G substitution has been shown to decrease shedding of S1 from spike on virions, consistent with increased stability of the pre-fusion conformation 13, 15. The virus has evolved in the human host during the pandemic 9, 10, 11 and we and others have demonstrated that the predominant D614G substitution, located in a monomer-monomer interface of the spike trimer, increases its propensity to adopt the open conformation that is competent to bind receptor 12, 13, 14. Its function is to bind the host receptor ACE2 and mediate the subsequent membrane fusion required for cell entry 1, 2, 3, 4, 5, 6, 7, 8. 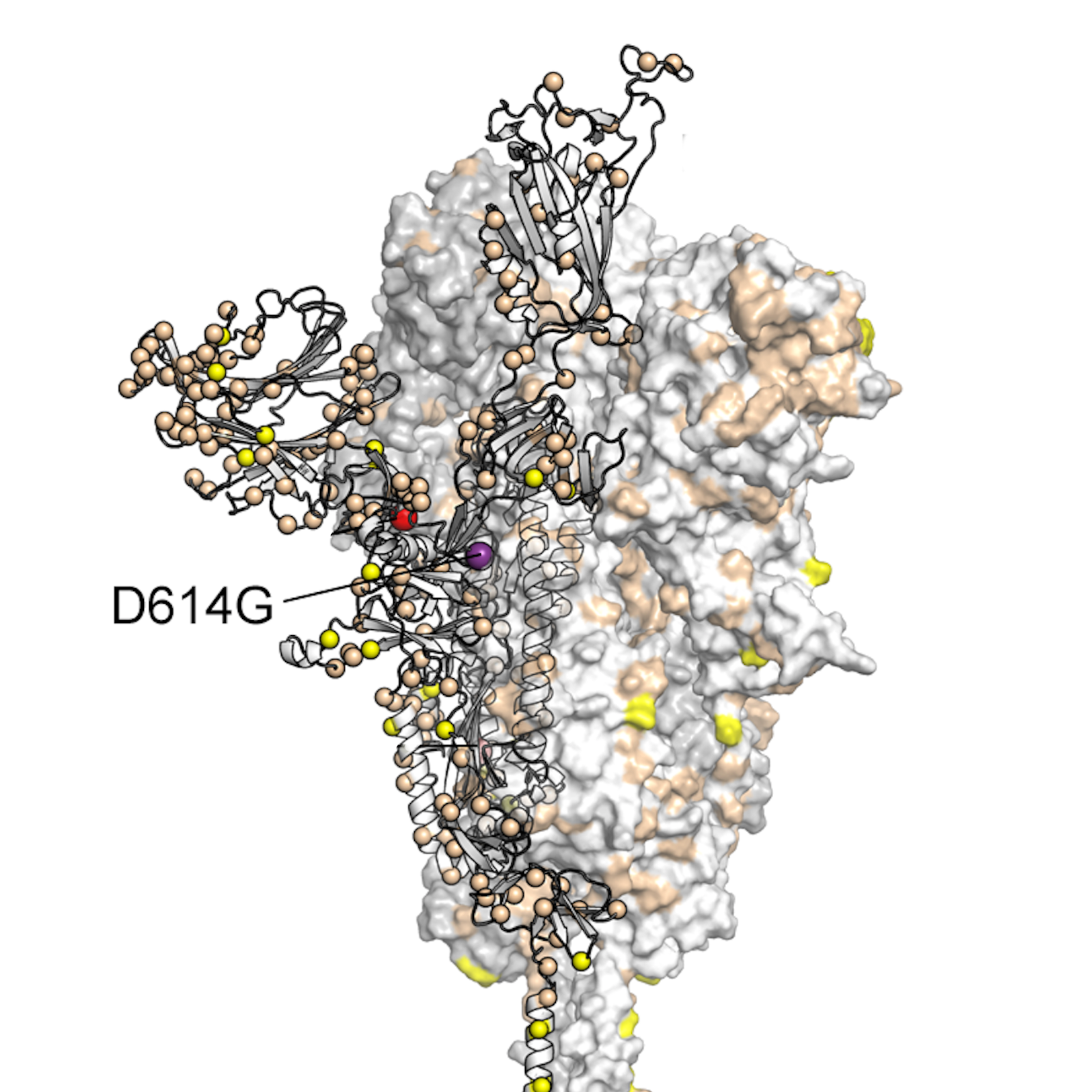
The SARS-CoV-2 spike glycoprotein is the major surface antigen of the virus. Our observations suggest ways these viruses have evolved to achieve greater transmissibility in humans. In the Beta variant spike, the presence of a new substitution, K417N (also observed in the Omicron variant), in combination with the D614G, stabilises a more open spike trimer, a conformation required for receptor binding. This feature is linked to the acquisition of a more basic substitution at the S1-S2 furin site (also observed for the variants of concern Delta, Kappa, and Omicron) which allows for near-complete cleavage. The spike from the Alpha variant is more stable against disruption upon binding ACE2 receptor than all other spikes studied. Each variant spike has acquired other key changes in structure that likely impact virus pathogenesis. This substitution confers tighter ACE2 binding, dependent on the common earlier substitution, D614G. Spikes of both variants have the same mutation, N501Y, in the receptor-binding domains. 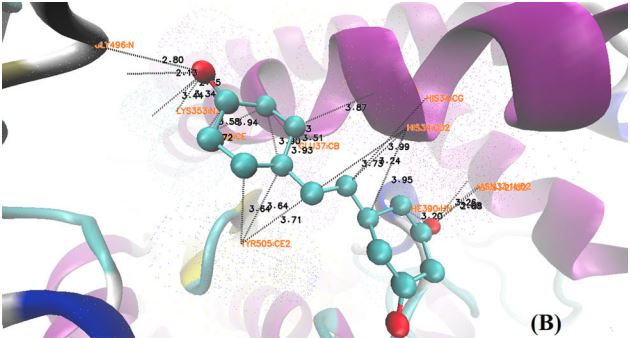 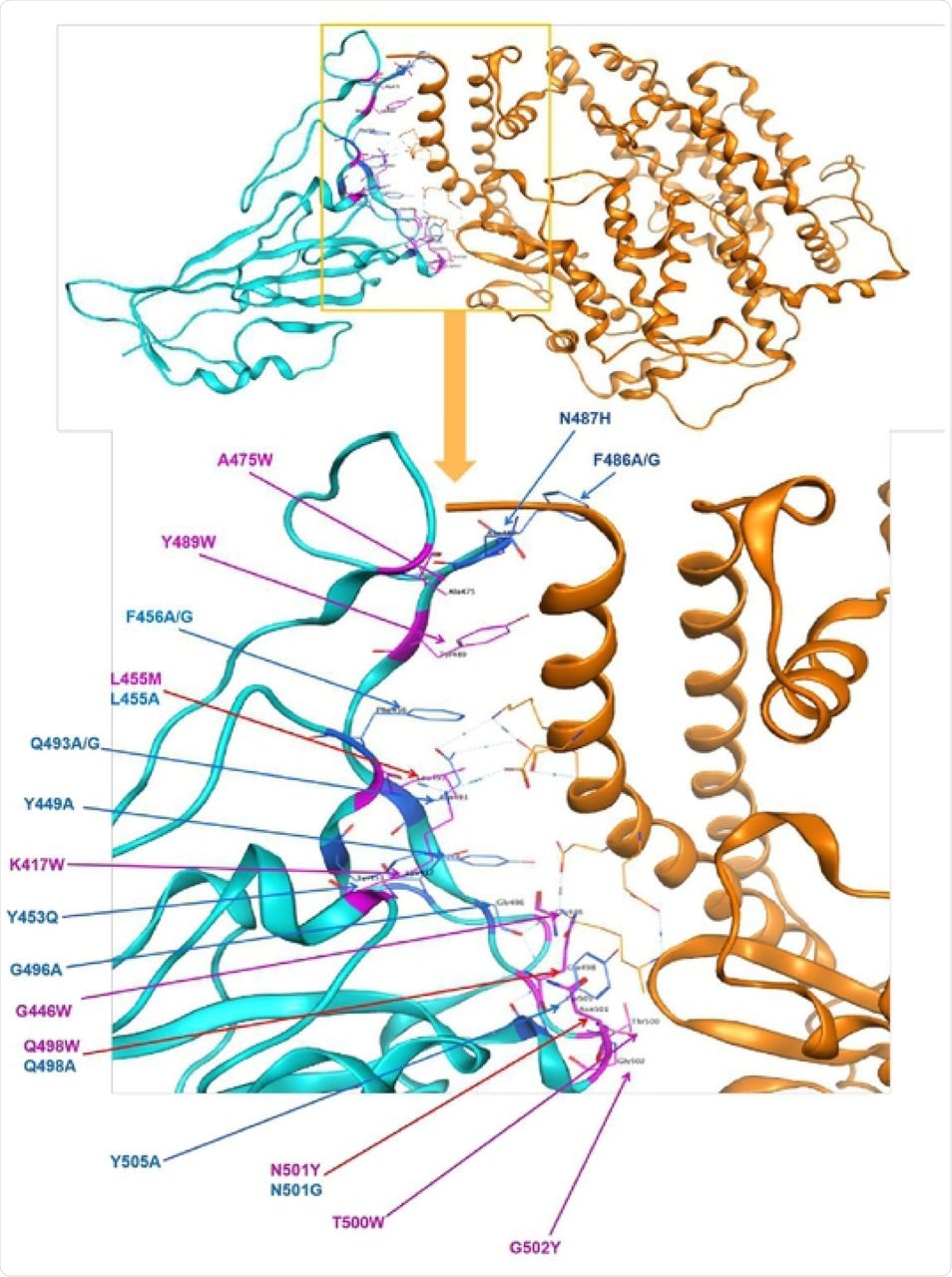
We have examined the structure and receptor binding properties of spike proteins from the B.1.1.7 (Alpha) and B.1.351 (Beta) variants to better understand the evolution of the virus in humans. Recently emerged variants of SARS-CoV-2 contain in their surface spike glycoproteins multiple substitutions associated with increased transmission and resistance to neutralising antibodies.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
